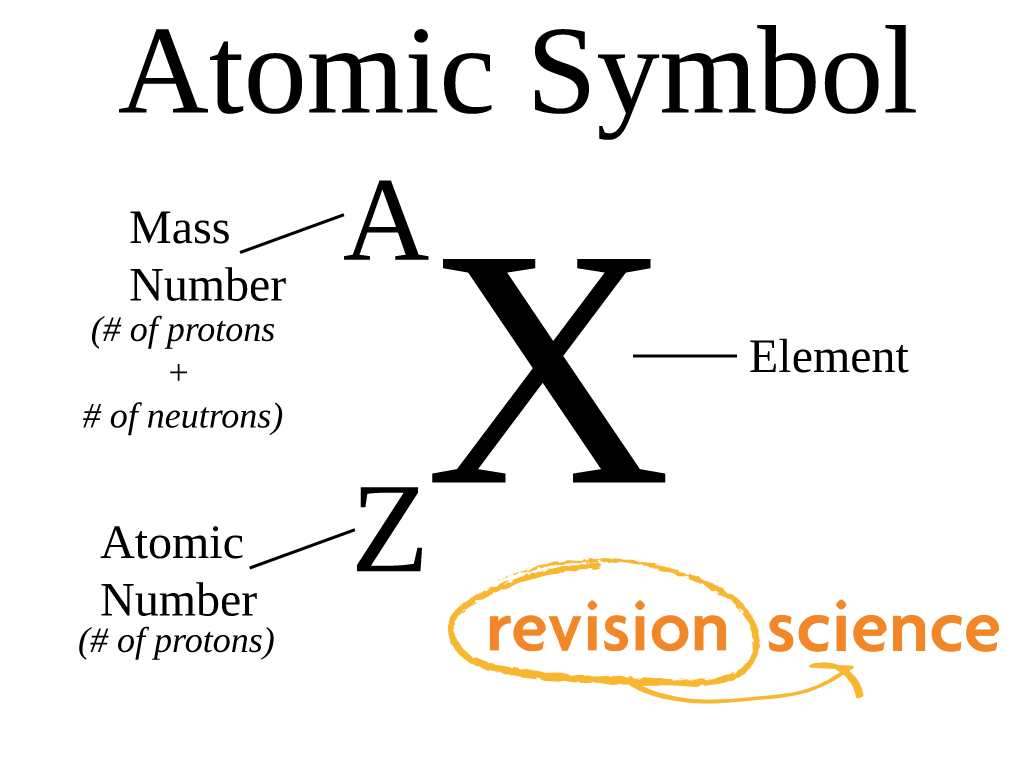
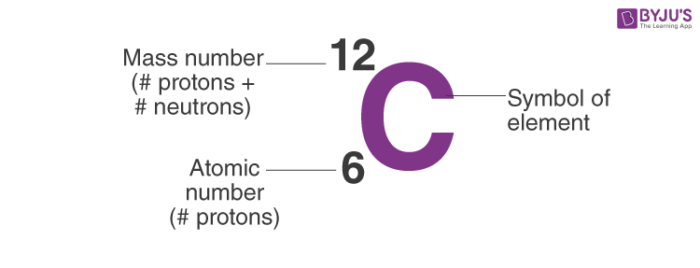
1 Revision - Atoms Atomic number, Z, number of protons in the nucleus. Atomic symbol, X, one or two letter code – uniquely related to atomic number. Mass. - ppt download

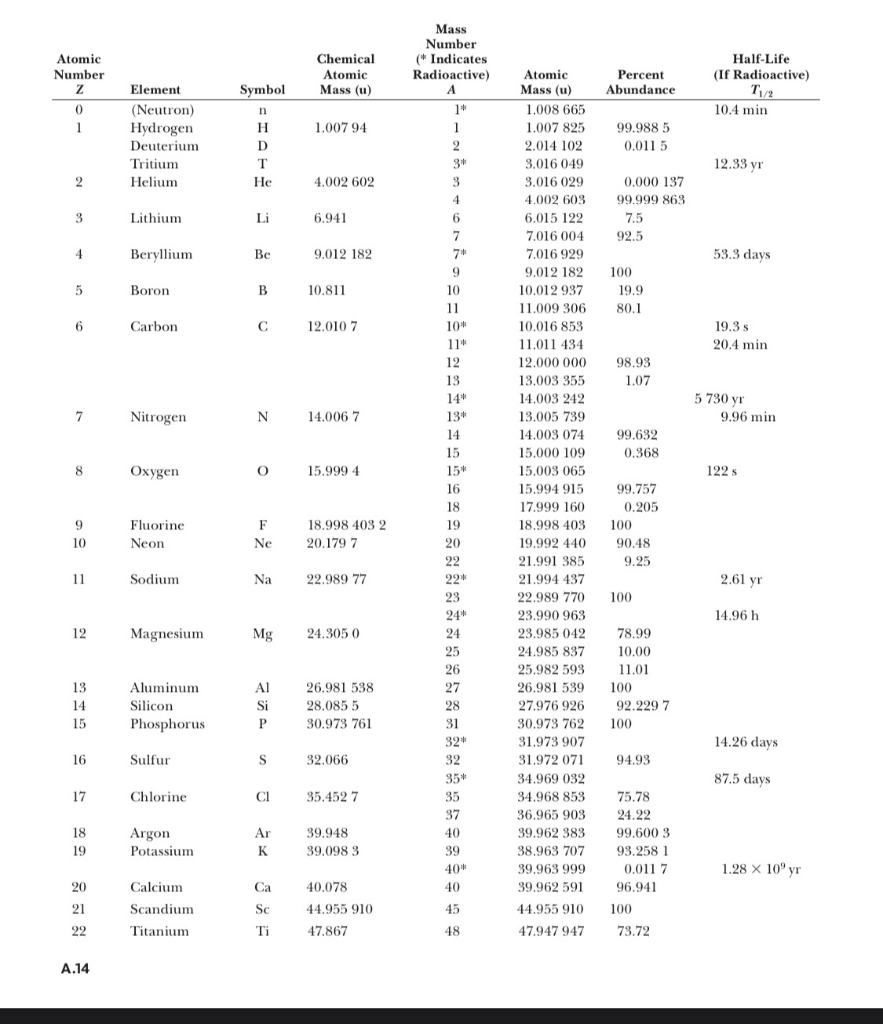
1 mass p ≈ mass n ≈ 1840 x mass e -. 2 Atomic number (Z) = number of protons in nucleus Mass number (A) = number of protons + number of neutrons = atomic. - ppt download
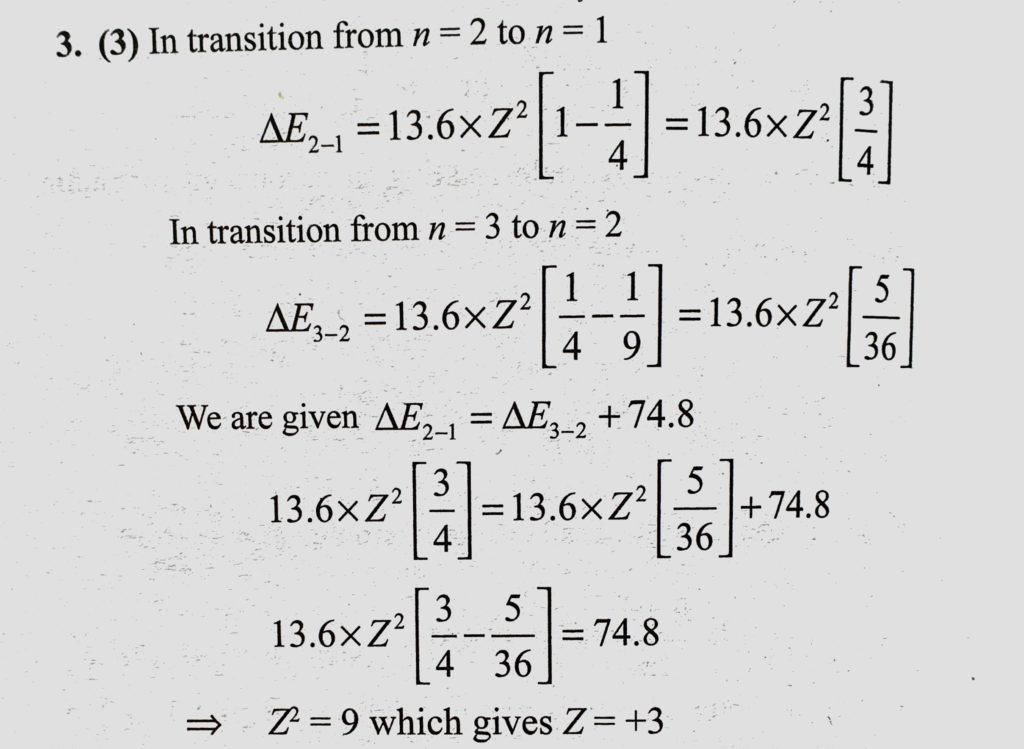
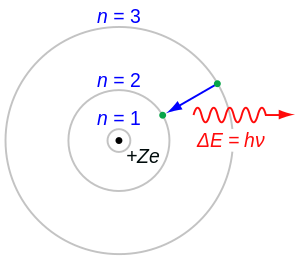
Consider a hydrogen-like ionized atom with atomic number Z with a single electron. In the emission spectrum of this atom, the photon emitted in the n = 2 to n = 1