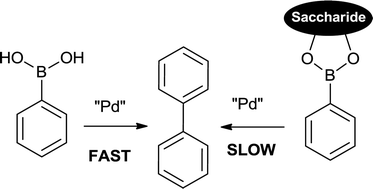
Suzuki homo-coupling reaction based fluorescent sensors for monosaccharides - RSC Advances (RSC Publishing)
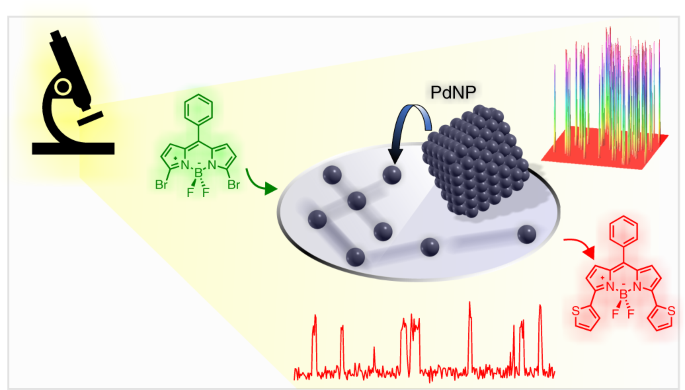
Real-time fluorescence imaging of a heterogeneously catalysed Suzuki–Miyaura reaction | Nature Catalysis

Catalysts | Free Full-Text | Rapid Sequentially Palladium Catalyzed Four-Component Synthesis of Novel Fluorescent Biaryl-Substituted Isoxazoles
![The synthesis of fluorescent benzofuro[2,3-c]pyridines via palladium-catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing) The synthesis of fluorescent benzofuro[2,3-c]pyridines via palladium-catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing)](https://pubs.rsc.org/image/article/2020/qo/c9qo01491h/c9qo01491h-s1_hi-res.gif)
The synthesis of fluorescent benzofuro[2,3-c]pyridines via palladium-catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing)
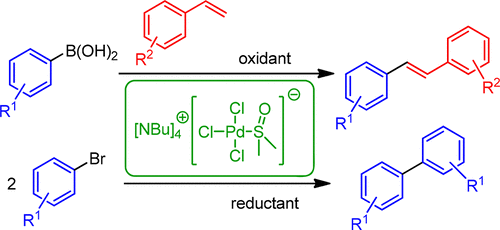
Oxidative and Reductive Cross-Coupling Reactions Catalyzed by an Anionic “Ligandless” Palladium Complex,Organic Process Research & Development - X-MOL
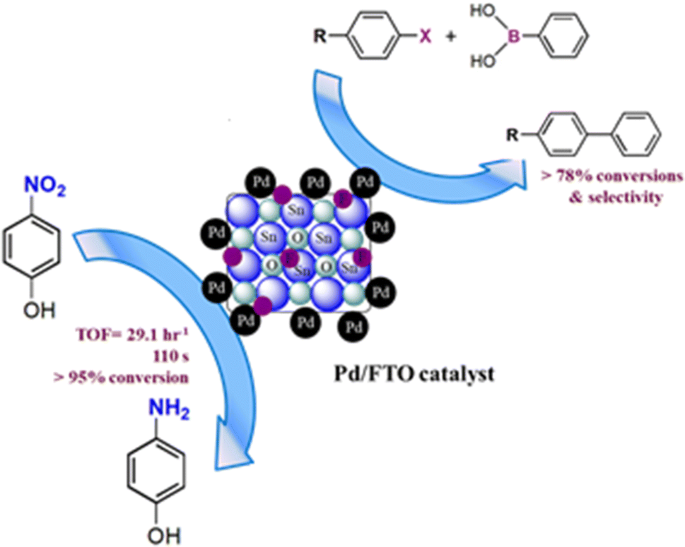
Palladium nanoparticles supported on fluorine-doped tin oxide as an efficient heterogeneous catalyst for Suzuki coupling and 4-nitrophenol reduction | SpringerLink

Palladium‐Catalyzed Coupling Reactions for the Functionalization of BODIPY Dyes with Fluorescence Spanning the Visible Spectrum - Rohand - 2006 - European Journal of Organic Chemistry - Wiley Online Library

New trends in the cross-coupling and other catalytic reactions in: Pure and Applied Chemistry Volume 89 Issue 10 (2017)

Highly selective Palladium-catalyzed Suzuki coupling reaction toward chlorine-containing electroluminescence polymers - ScienceDirect

A near-infrared and colorimetric fluorescent probe for palladium detection and bioimaging - ScienceDirect
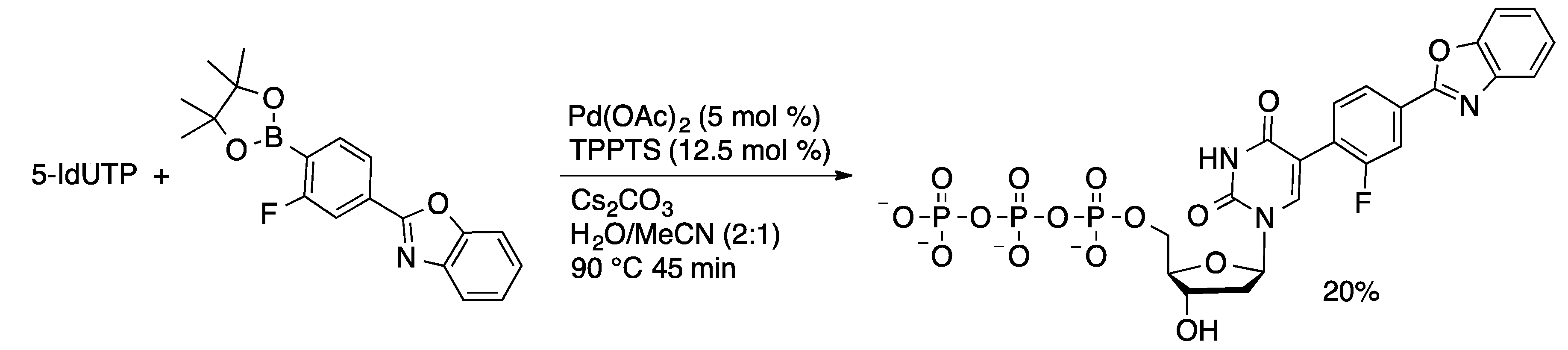
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

Suzuki Cross‐Coupling Reaction with Genetically Encoded Fluorosulfates for Fluorogenic Protein Labeling - Zhao - - Chemistry – A European Journal - Wiley Online Library
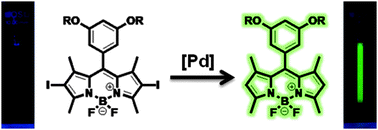
A fluorescence turn-on sensor for the detection of palladium ions that operates through in situ generation of palladium nanoparticles - Chemical Communications (RSC Publishing)

Proposed mechanism for the palladium pincer-catalyzed carbonylative... | Download Scientific Diagram
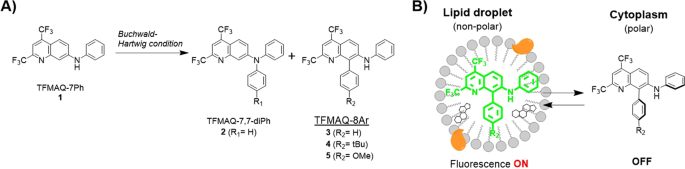
Selective synthesis of substituted amino-quinoline derivatives by C-H activation and fluorescence evaluation of their lipophilicity-responsive properties | Scientific Reports
![Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402017303848-fx1.jpg)
Palladium-catalyzed synthesis and fluorescence study of 2,3-diaryl-5-ethynylbenzo[e]indoles - ScienceDirect

A highly selective fluorescent probe for the detection of palladium(II) ion in cells and aqueous media | SpringerLink
![Synthesis of β‐Benzo[b]thienyldehydrophenylalanine Derivatives by One‐Pot Palladium‐Catalyzed Borylation and Suzuki Coupling (BSC) and Metal‐Assisted Intramolecular Cyclization – Studies of Fluorescence and Antimicrobial Activity - Abreu - 2005 ... Synthesis of β‐Benzo[b]thienyldehydrophenylalanine Derivatives by One‐Pot Palladium‐Catalyzed Borylation and Suzuki Coupling (BSC) and Metal‐Assisted Intramolecular Cyclization – Studies of Fluorescence and Antimicrobial Activity - Abreu - 2005 ...](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/2b5bc92b-a422-45aa-919a-1fdee01dc82e/mfig000.gif)
Synthesis of β‐Benzo[b]thienyldehydrophenylalanine Derivatives by One‐Pot Palladium‐Catalyzed Borylation and Suzuki Coupling (BSC) and Metal‐Assisted Intramolecular Cyclization – Studies of Fluorescence and Antimicrobial Activity - Abreu - 2005 ...
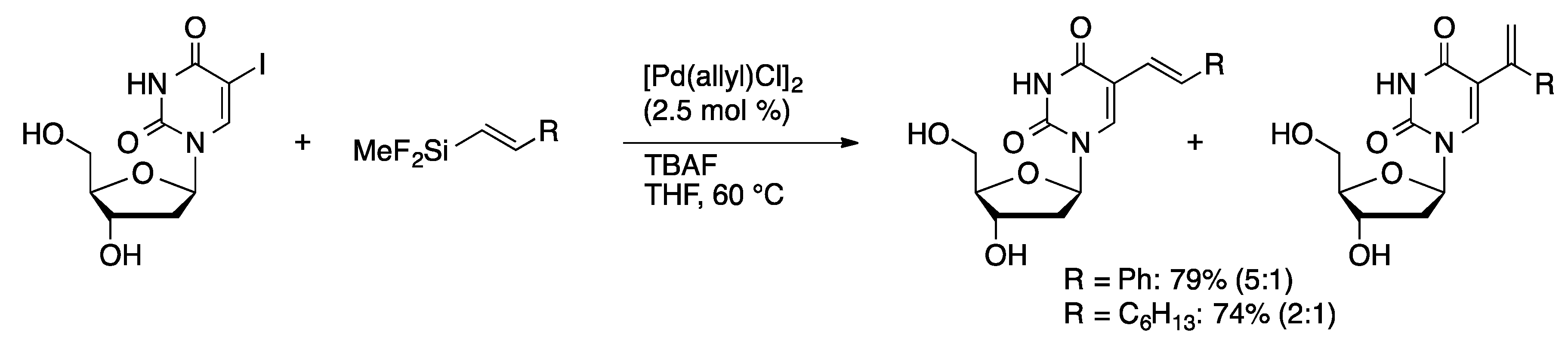
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML




