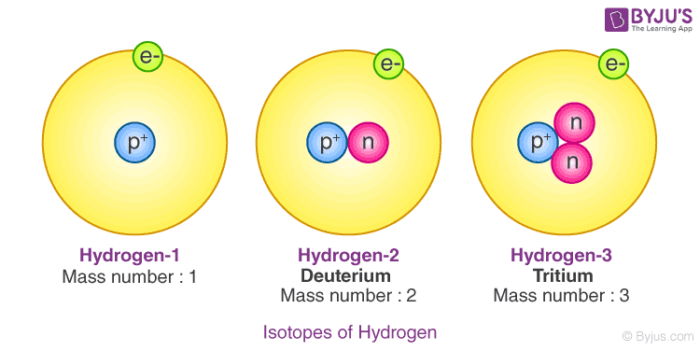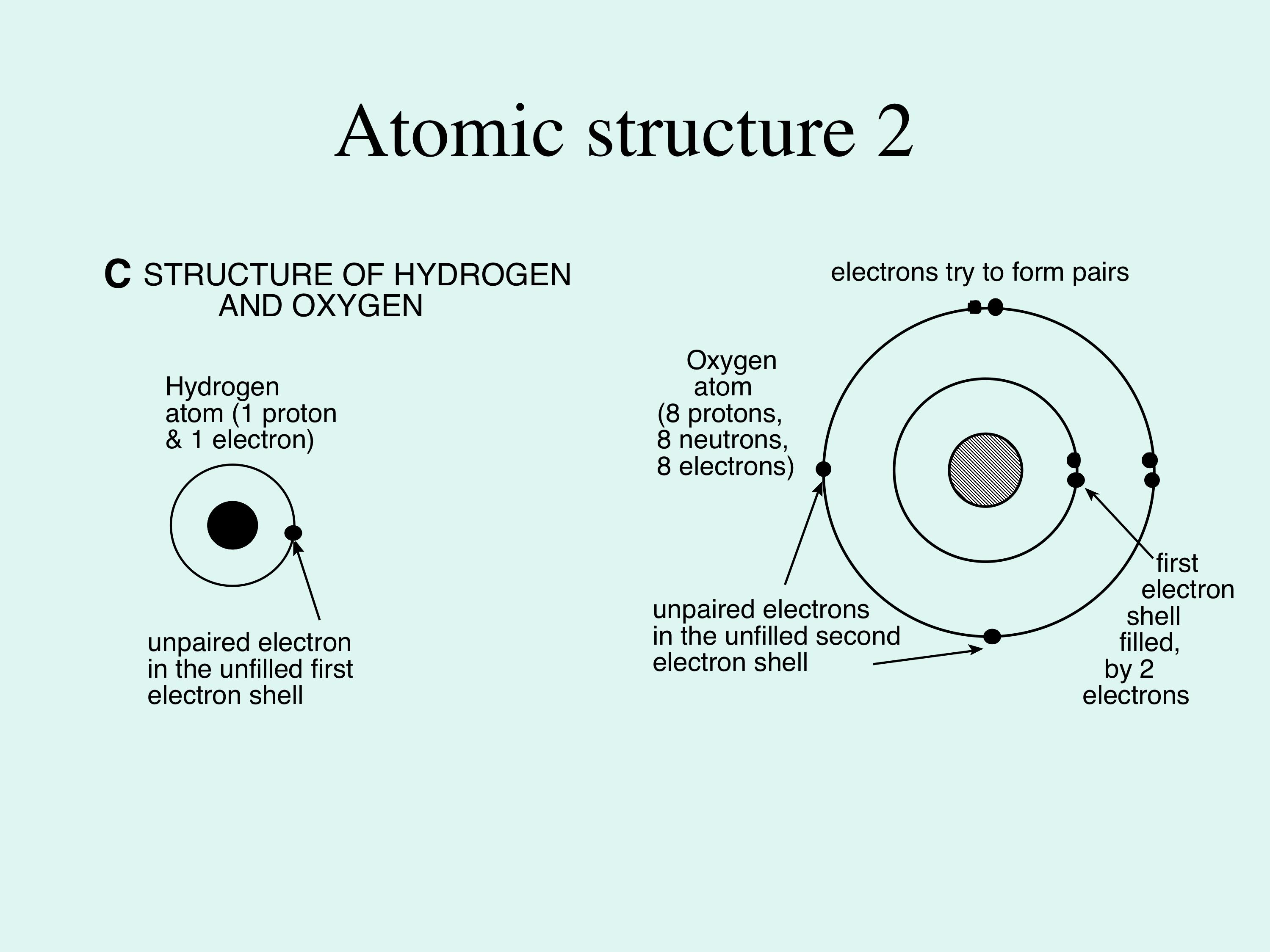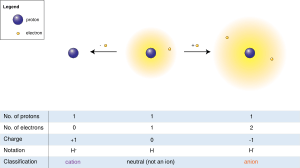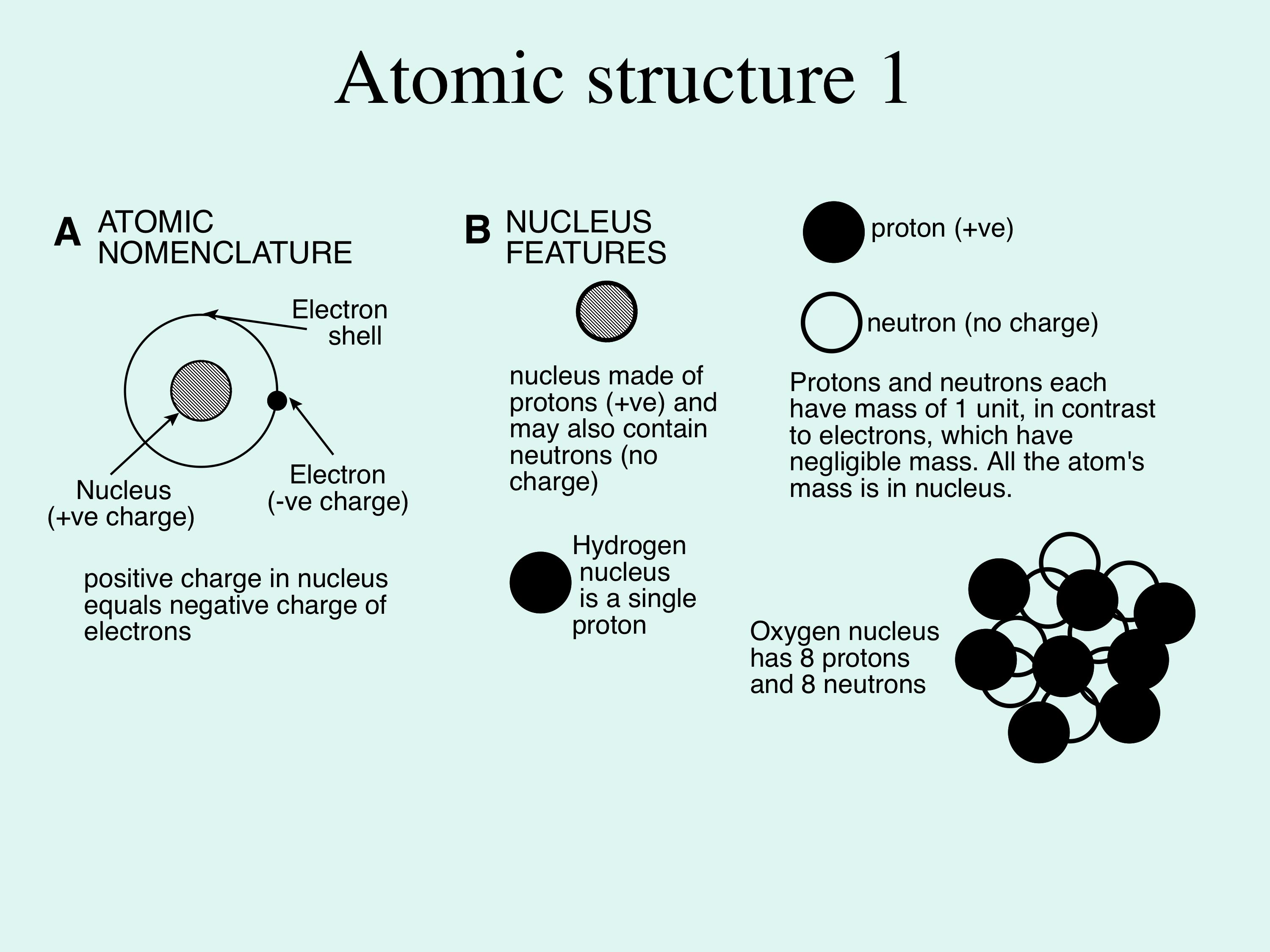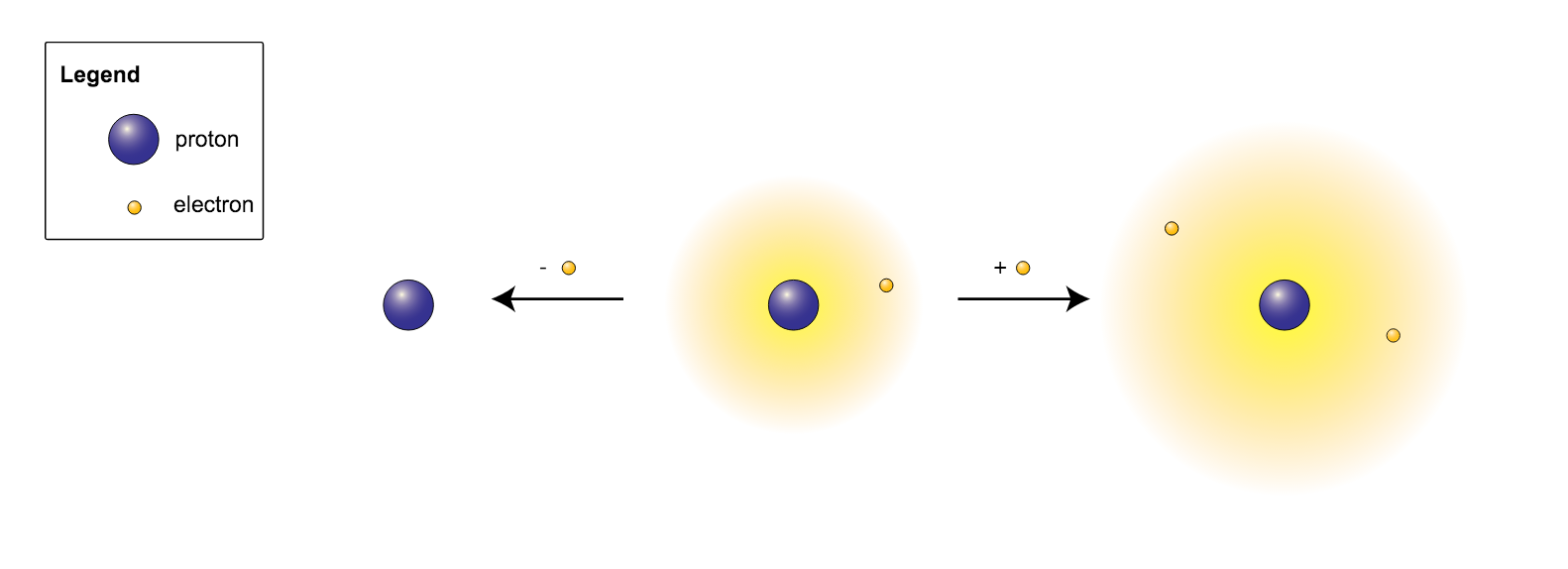
When Atoms Change- Ions. What happens if the number of electrons change??? If an atom gains electrons… 1. The charge becomes negative. (There are more. - ppt download

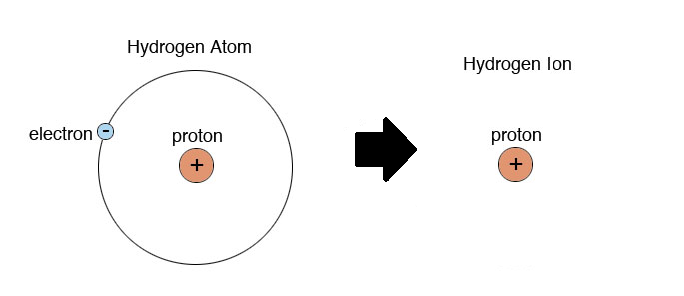
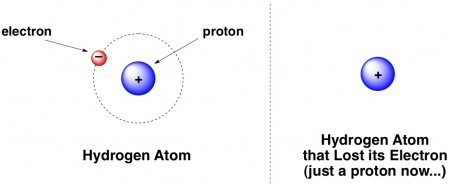
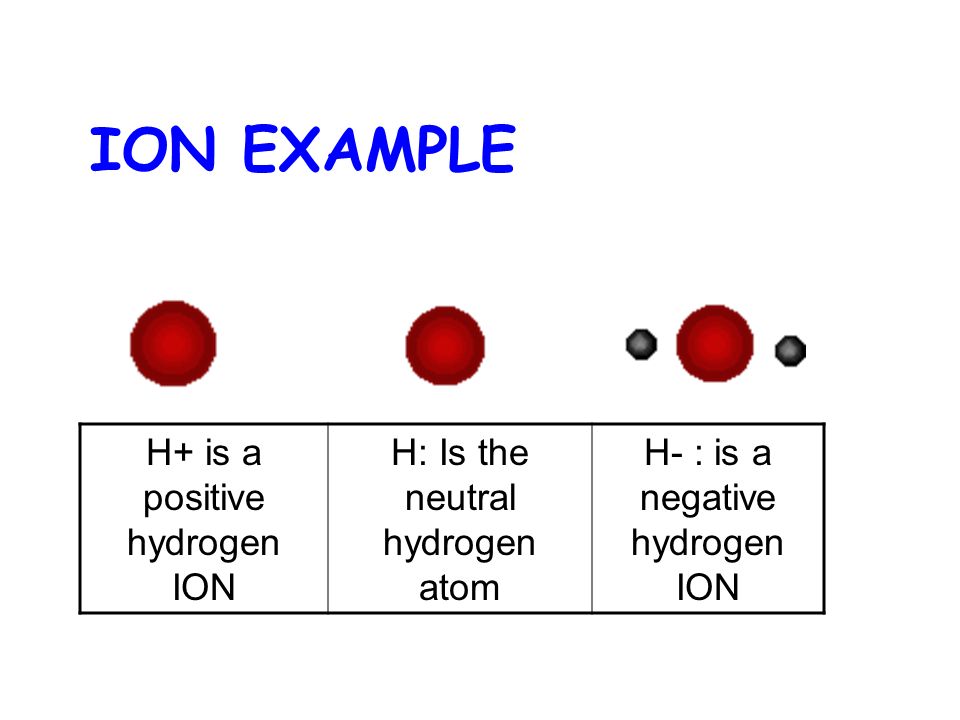
Figure 3 from Positively charged Hydrogen atom(H+) different from Proton (p+): Proposed model for constituent particles in positively charged Hydrogen atom(H+). | Semantic Scholar

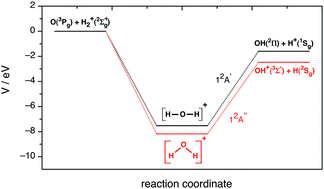
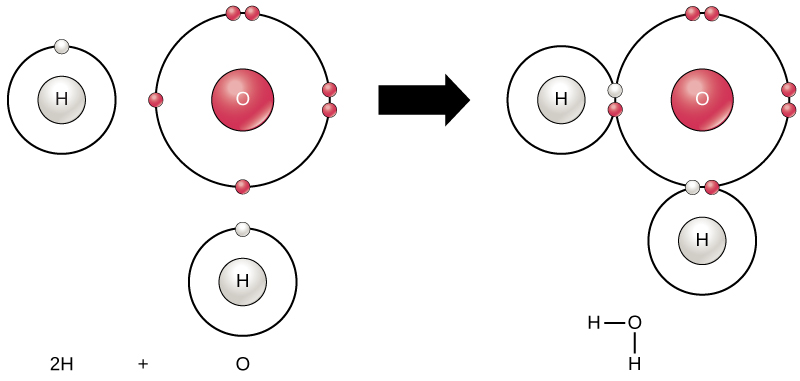
The Dissociation of Water Molecules - Chapter 3: Water and the Fitness of the Environment by Josh Birkett