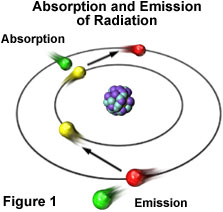
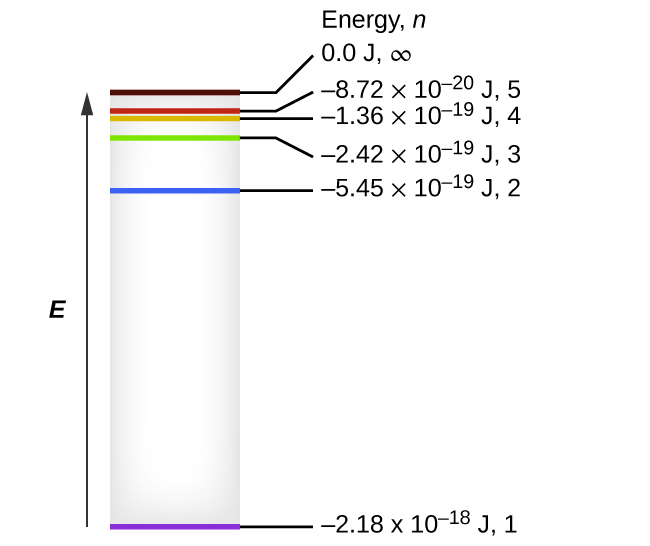
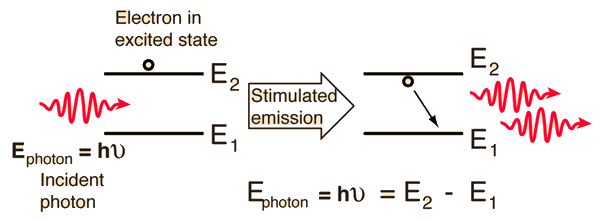
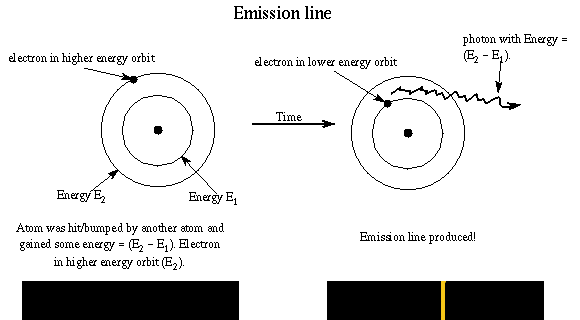
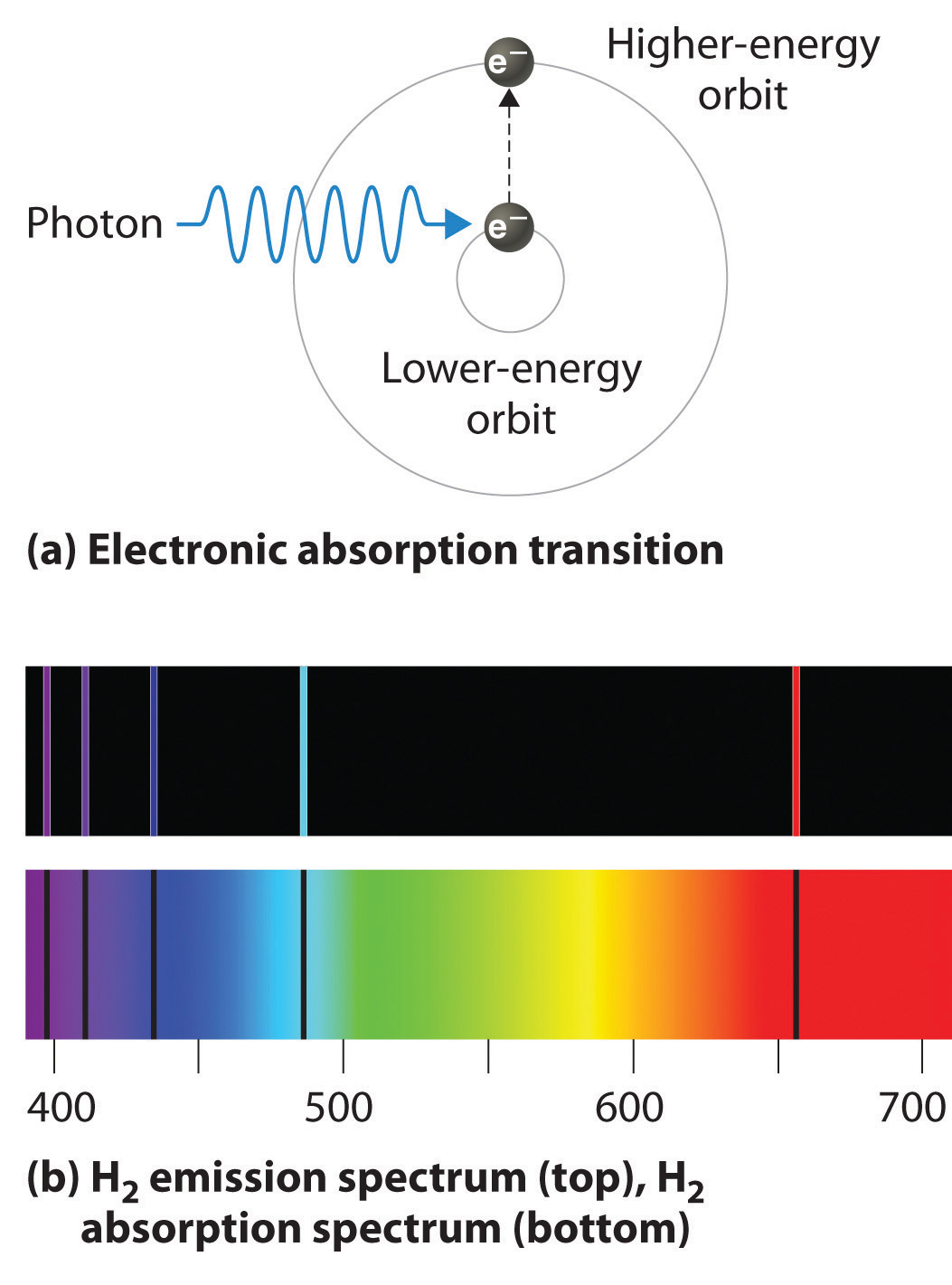
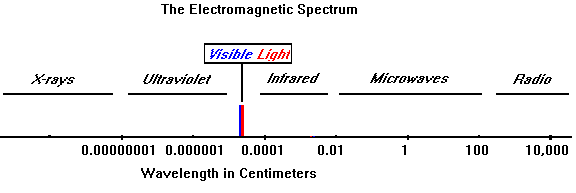
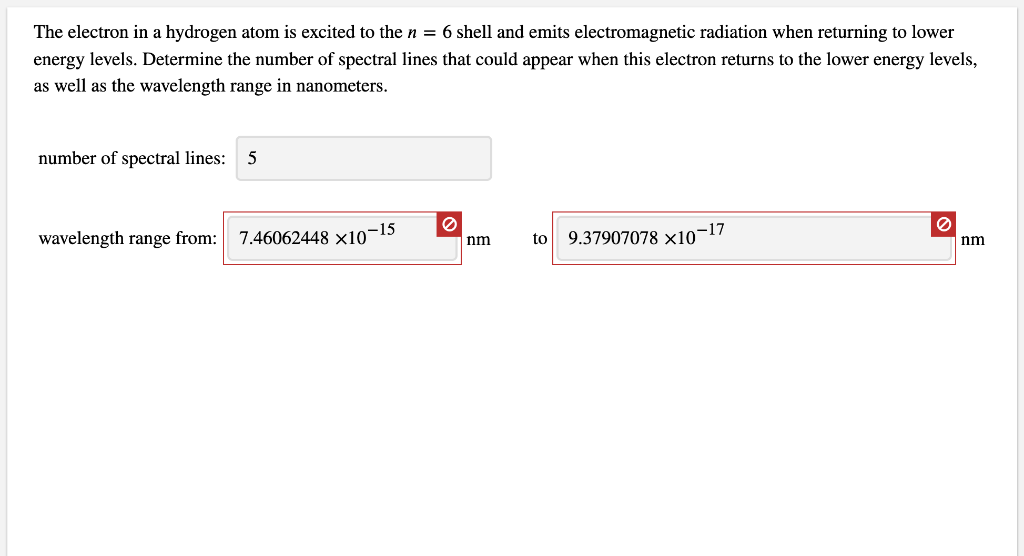
Absorption of radiation, Spontaneous emission and Stimulated emission – Physics and Radio-Electronics

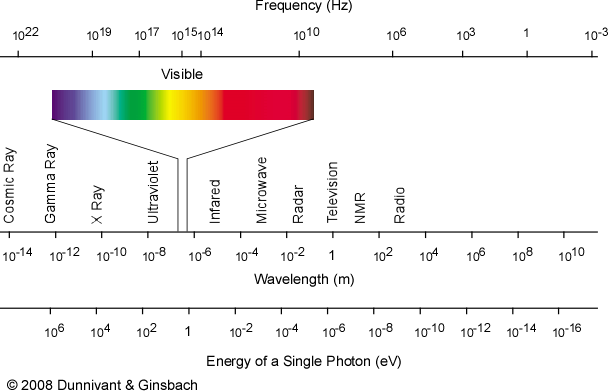
medical imaging using electromagnetic radiation communication uses properties dangers EM waves radio waves microwaves infrared radiation visible light ultraviolet X-rays gamma rays EM spectrum igcse/gcse 9-1 Physics revision notes