
Figure 1 from Solubility of ThO2·xH2O(am) and the formation of ternary Th(IV) hydroxide-carbonate complexes in NaHCO3-Na2CO3 solutions containing 0−4 M NaCl | Semantic Scholar
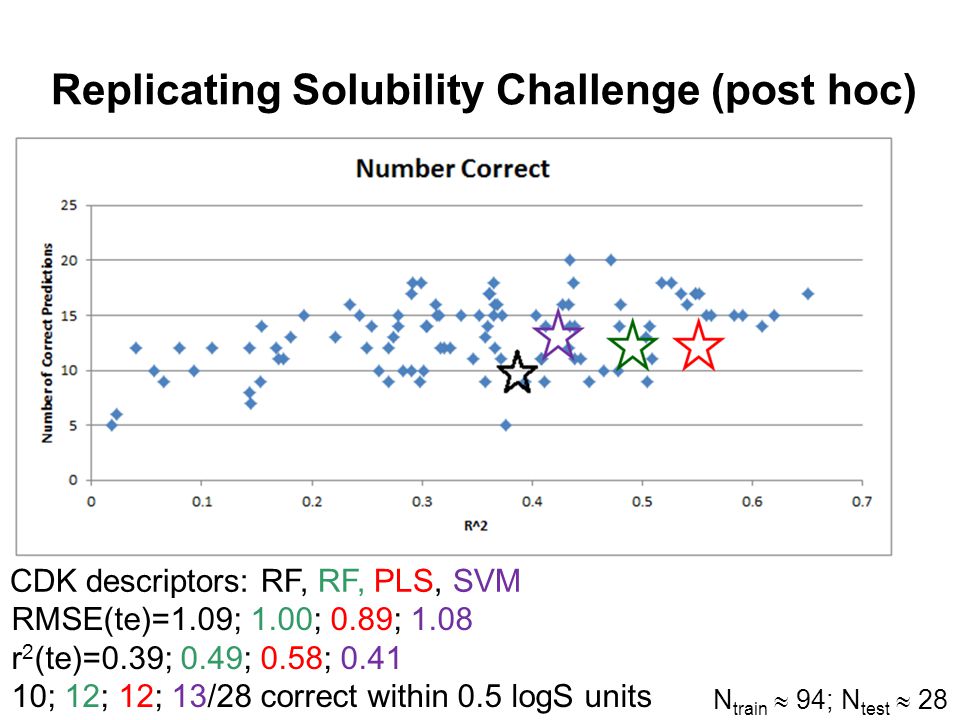
Quantum Chemical and Machine Learning Approaches to Property Prediction for Druglike Molecules Dr John Mitchell University of St Andrews. - ppt download

Solubility and its component concentrations of Np(IV) at 0.5 M NaClO 4... | Download Scientific Diagram
Figure A8. Vox Machinarum predictions plotted against Avdeef's average... | Download Scientific Diagram

Logarithmic Partition Coefficients of PDA Ligands (log P) in the 0.5 M... | Download Scientific Diagram
![Pu solubility in the presence of [C]tot = 0.16 mol·dm⁻³ in I = 0.5 and... | Download Scientific Diagram Pu solubility in the presence of [C]tot = 0.16 mol·dm⁻³ in I = 0.5 and... | Download Scientific Diagram](https://www.researchgate.net/publication/350893910/figure/fig3/AS:1013085980598285@1618549928442/Pu-solubility-in-the-presence-of-Ctot016moldm-in-I05-and-20moldm-NaNO3-as-a.png)
Pu solubility in the presence of [C]tot = 0.16 mol·dm⁻³ in I = 0.5 and... | Download Scientific Diagram

The role of pH and dose/solubility ratio on cocrystal dissolution, drug supersaturation and precipitation - ScienceDirect
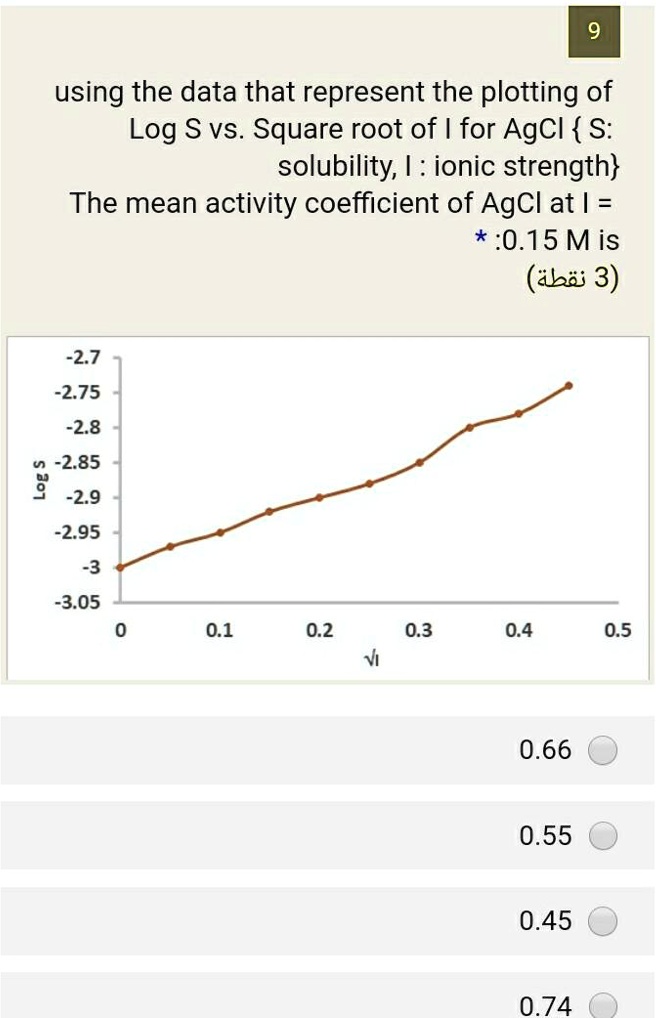
SOLVED:using the data that represent the plotting of Log S vs Square root of for AgCl { S: solubility, I : ionic strength} The mean activity coefficient of AgCl at :0.15 Mis (
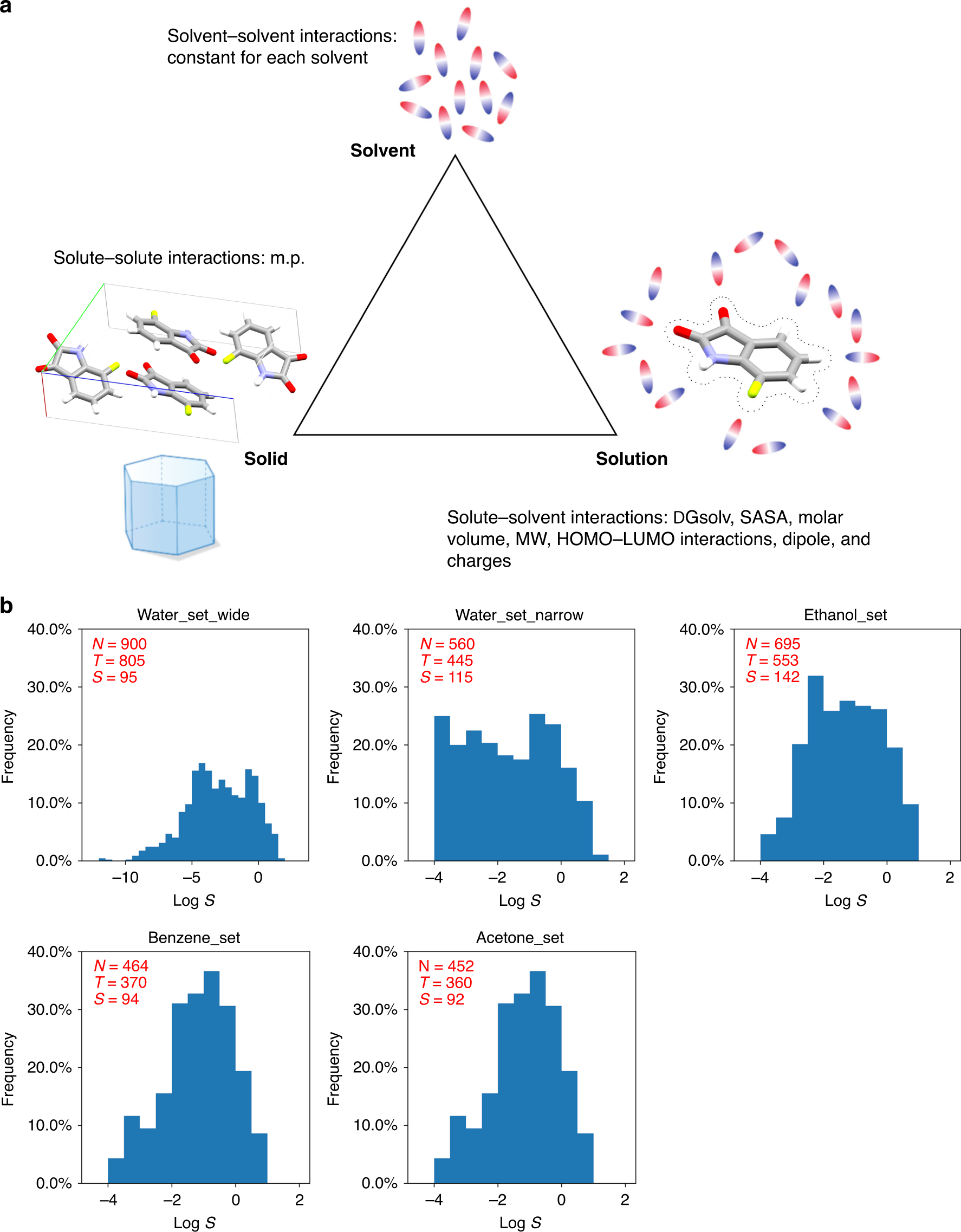
Machine learning with physicochemical relationships: solubility prediction in organic solvents and water | Nature Communications

Figure 2 from Solubility of ThO2·xH2O(am) and the formation of ternary Th(IV) hydroxide-carbonate complexes in NaHCO3-Na2CO3 solutions containing 0−4 M NaCl | Semantic Scholar
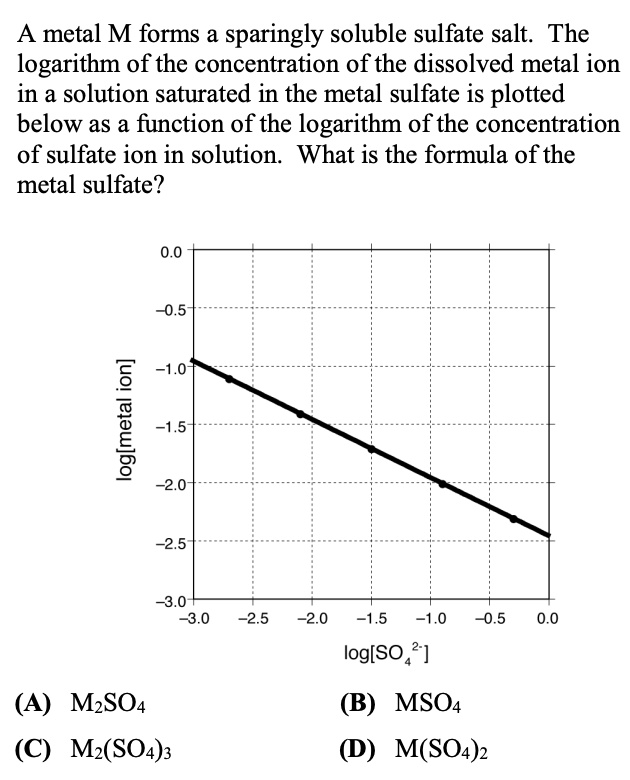
SOLVED:A metal M forms sparingly soluble sulfate salt: The logarithm of the concentration of the dissolved metal ion in a solution saturated in the metal sulfate is plotted below as a function
![What is the pH of a saturated solution of X(OH)3 will be if its solubility product = 2.7 × 10^-43 ?[Given: log 3 = 0.5, log 2 = 0.3] What is the pH of a saturated solution of X(OH)3 will be if its solubility product = 2.7 × 10^-43 ?[Given: log 3 = 0.5, log 2 = 0.3]](https://d2rrqu68q7r435.cloudfront.net/images/7984587/97259092-21e7-413a-aa0c-7fee11193a62.jpg)









![Solubility of Zr(IV) in 0.5 mol·dm⁻³ NaNO3 as a function of [C]tot... | Download Scientific Diagram Solubility of Zr(IV) in 0.5 mol·dm⁻³ NaNO3 as a function of [C]tot... | Download Scientific Diagram](https://www.researchgate.net/publication/315922876/figure/fig2/AS:961857066131456@1606336003242/Solubility-of-ZrIV-in-05moldm-NaNO3-as-a-function-of-Ctot-after-ultrafiltration.gif)

